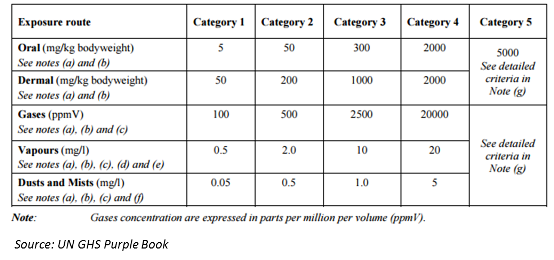
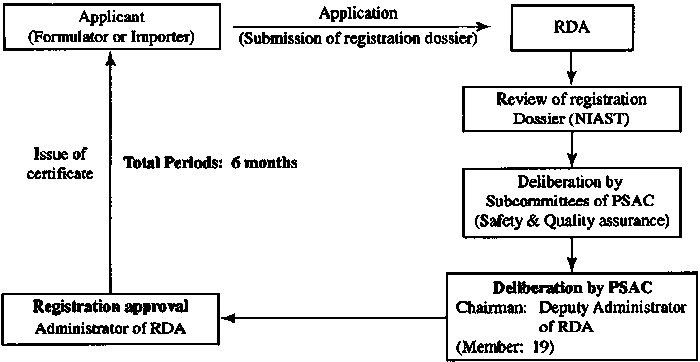
| Sr. No. |
TYPE / NAME OF THE STUDY |
EPA, OPPTS |
OECD |
EEC |
| ACUTE |
|
|
|
|
| 1 |
Acute oral rat |
870-1100 |
420/423/425 |
EU B.1 |
| 2 |
Acute oral mice |
870-1100 |
423 |
EU B.1 |
| 3 |
Acute dermal rat |
870-1200 |
402 |
EU B.3 |
| 4 |
Acute inhalation rat (Full / Limit) |
870-1300 |
403 |
EU B.2 |
| 5 |
Dermal irritation rabbit |
870-2500 |
404 |
EU B.4 |
| 6 |
Mucous membrane irritation rabbit |
870-2400 |
405 |
EU B.5 |
| 7 |
Skin sensitization - Buehler method |
870-2600 |
406 |
EU B.6 |
| 8 |
Skin sensitization - M & K |
870-2600 - |
406 |
EU B.6 - |
| 9 |
Local Lymph Node Assay in Mice |
|
429 |
|
| CHRONIC |
|
|
|
|
| 10 |
28 days oral rat |
870-3050 |
407 |
EU B.7 |
| 11 |
28 days dermal rat |
870-3200 |
410 |
EU B.9 |
| 12 |
28 days dermal rabbit |
870-3200 |
410 |
EU B.9 |
| 13 |
90 days oral rat |
870-3100 |
408 |
EU B.26 |
| 14 |
14 days inhalation rat |
- |
412 |
- |
| 15 |
28 days inhalation rat |
- |
412 |
EU B.8 |
| 16 |
90 days inhalation rat |
870-3465 |
413 |
EU B.29 |
| GENOTOXICITY |
|
|
|
|
| 17 |
Ames test |
870-5100 |
471 |
B.14 |
| 18 |
In vitro mammalian gene mutation test |
870-5300 |
476 |
B.17 |
| 19 |
In vitro human lymphocyte |
870-5375 |
473 |
B.10 |
| 20 |
Chromosomal aberration in mice |
870-5385 |
475 |
B.11 |
| 21 |
Micronucleus test in mice |
870-5395 |
474 |
B.12 |
| REPRODUCTION |
|
|
|
|
| 22 |
Teratology /Prenatal development in rats |
830-3200 |
414 |
B.31 |
| 23 |
Teratology /Prenatal development in rabbits |
830-3200 |
414 |
B.31 |
| 24 |
One generation in rats |
- |
415 |
B.34 |
| 25 |
Two generation in rats |
870.63 |
416 |
B.35 |
| 26 |
Development Neurotoxicity in Rats |
- |
422 |
- |
| CARCINOGENICITY |
|
|
|
|
| 27 |
Carcinogenicity in rats |
870.42 |
451 |
B.32 |
| 28 |
Carcinogenicity in mouse |
870.42 |
451 |
B.32 |
| 29 |
Combined chronic carcinogenicity in rats |
870.43 |
453 |
B.33 |
| 30 |
Chronic toxicity in rats |
870.41 |
452 |
B.30 |
| AQUATIC |
|
|
|
|
| 31 |
Alga growth inhibition test |
- |
201 |
C.3 |
| 32 |
48 h. Acute immobilization Daphnia magna |
|
202 |
C.2 |
| 33 |
Acute fish tox-Common Carp (Semi static) |
|
203 |
C.1 |
| 34 |
Acute fish tox- Rainbow trout (Semi static) |
|
203 |
C.1 |
| 35 |
Reproduction test - Daphnia magna |
|
211 |
C.20 |
| 36 |
A.I. Method validation for Ecotox Studies |
|
- |
- |
| TERRESTRIAL |
|
|
|
|
| 37 |
Avian dietary toxicity test - Japanese quail |
--------- |
205 |
- |
| 38 |
Avian dietary toxicity test - Mallard duck |
|
205 |
- |
| 39 |
Avian reproduction test - Japanese quail |
|
206 |
- |
| 40 |
Avian acute oral toxicity - Japanese quail |
|
Setac |
- |
| 41 |
Acute toxicity - earthworm (Eisenia foetida) Earthworm reproduction toxicity/ Earthworm |
|
207 |
C.8 |
| 42 |
subchronic toxicity |
|
222 |
- |
| 43 |
Acute toxicity-honeybee oral(Apis mellifera) |
|
213 |
C.16 |
| 44 |
Acute tox honeybee-contact(Apis mellifera) |
|
214 |
C.17 |
| 45 |
Acute toxicity to honeybee - oral & contact |
|
- |
EPPO. |